Quick Summary
Click here for Price and Turnaround Time
Phenotype: Cats affected by hypertrophic cardiomyopathy (HCM) are at risk of sudden cardiac death. Ragdoll cats with two copies of the disease-associated mutation are at high risk of developing severe signs of HCM, usually between 1-2 years of age, and have a greater likelihood of early cardiac death. Cats that carry one copy of the mutation are not likely to show signs of the disease and may live a normal lifespan.
Mode of Inheritance: Autosomal dominant (incomplete penetrance)
Alleles: N = Normal, HCMrd = hypertrophic cardiomyopathy-associated mutation
Breeds appropriate for testing: Ragamuffin, Ragdoll and related outcrosses. This variant has also been detected in the American Bobtail (Longhair), Highlander and Munchkin.
Explanation of Results:
- Cats with N/N genotype are not predicted to be at increased risk of HCM and cannot transmit this hypertrophic cardiomyopathy-associated mutation to their offspring.
- Cats with N/HCMrd genotype may be at increased risk of HCM. They will transmit this hypertrophic cardiomyopathy-associated mutation to 50% of their offspring.
- Cats with HCMrd/HCMrd genotype are at high risk of developing HCM, a potentially fatal condition.
Sample Collection:
Cat DNA tests are carried out using cells collected from your cat's cheeks and gums using household cotton swabs.
The cat DNA submission form with instructions, pricing, and a place to tape the cotton swabs can be printed from your home computer after signing up for a MyVGL account and placing an order. Test kits are not mailed.
Step-By-Step Instructions:
1.
 Purchase regular household cotton swabs for cat DNA collection (the cotton swabs can be purchased at a pharmacy or drug store)
Purchase regular household cotton swabs for cat DNA collection (the cotton swabs can be purchased at a pharmacy or drug store)
2.
 Use both ends of the two cotton swabs for a total of four swabs.
Use both ends of the two cotton swabs for a total of four swabs.
3.
 Collect the DNA sample by swabbing the cheek and gums of the cat.
Collect the DNA sample by swabbing the cheek and gums of the cat.
4.
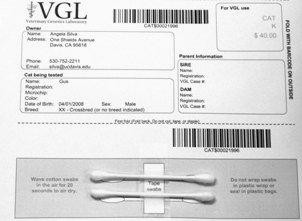 After swabbing the cheek and gums, tape the cotton swabs to the bar-coded submission form printed from your MyVGL account.
After swabbing the cheek and gums, tape the cotton swabs to the bar-coded submission form printed from your MyVGL account.
Hypertrophic cardiomyopathy is the most common cardiac disease in cats. Affected cats are at risk of sudden cardiac death due to defects that produce increased left ventricular heart muscle thickness. In Ragdolls, the condition is inherited due to breed specific mutations in the cardiac myosin binding protein C gene (MYBPC3).
The Ragdoll HCM mutation, known as R820W, is a single base pair change in MYBPC3 that is thought to alter the shape and function of this essential protein for normal heart muscle development. The same R820W mutation has been found to be associated with HCM and left ventricular non-compaction in humans (see reference below, Ripoll et al. 2010). Recent studies show cats that are heterozygous (1 copy) for the mutation are not likely to show signs of the disease and may live a normal lifespan. Homozygous (2 copies) cats for the mutation are at high risk of developing severe HCM signs, usually between 1-2 years of age and have a greater likelihood of early cardiac death. Infrequently, homozygous cats do not show clinical signs of HCM.
Breedings between 2 heterozygous cats are expected to produce 25% high risk kittens. It is not recommended to use cats homozygous for the R820W mutation in a breeding program.
Testing for the Ragdoll HCM mutation to helps owners and breeders identify the Ragdoll HCM status of their cats.



